Epithelial–Mesenchymal Transition in Cancer Progression: Biological Basis and Histopathological Implications
Main Article Content
Abstract
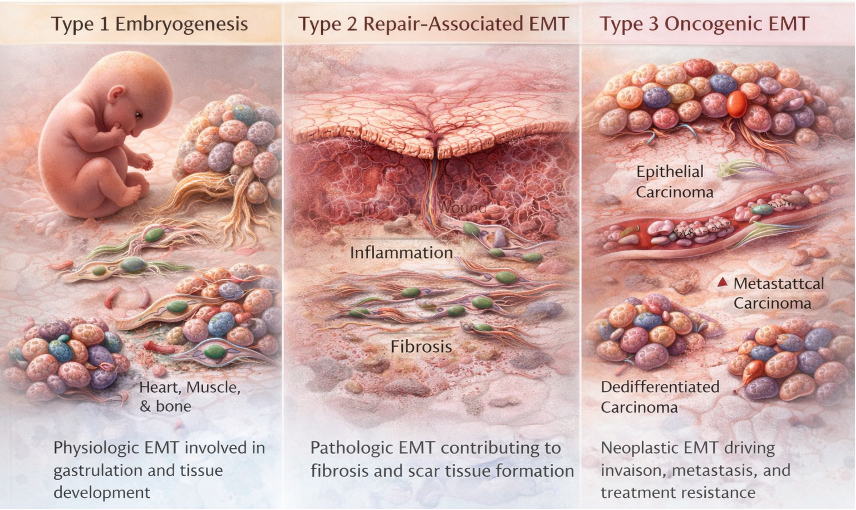
Background: Epithelial–mesenchymal transition (EMT) is a fundamental biologic process that plays a critical role in tumor invasion, metastasis, and dedifferentiation. While extensively investigated in molecular oncology, its relevance to routine surgical pathology practice remains underemphasized. Many morphologic alterations observed in daily histopathologic evaluation represent phenotypic manifestations of EMT, yet these findings are often interpreted descriptively without integration into a unified biologic framework.
Methods: This review aims to provide a practical overview of EMT from a surgical pathology perspective by correlating biologic mechanisms with histomorphologic features, immunohistochemical findings, and clinical implications across solid tumors.A narrative literature review was conducted using PubMed/MEDLINE databases to identify relevant studies published between 2020 and 2025. Keywords related to EMT, histopathology, tumor progression, and immunohistochemistry were applied. Selected articles were qualitatively synthesized with emphasis on clinicopathologic relevance and applicability to routine diagnostic practice.
Results: EMT is characterized by loss of epithelial differentiation, acquisition of mesenchymal traits, and increased cellular plasticity. Histologically, EMT correlates with recognizable features including loss of cellular cohesion, tumor budding, single-cell invasion, spindle cell transformation, and tumor dedifferentiation. Immunohistochemical alterations such as decreased E-cadherin expression and increased mesenchymal marker expression further support EMT-associated phenotypes. Across multiple organ systems, these morphologic changes are consistently associated with aggressive tumor behavior, metastatic potential, and therapeutic resistance.
Conclusion: EMT provides a unifying concept linking molecular cancer biology with routine histopathologic observations. Recognition of EMT-associated morphologic patterns may enhance prognostic assessment and strengthen clinicopathologic interpretation, reinforcing the essential role of histopathology in the era of precision oncology.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
References
Allgayer H, Mahapatra S, Mishra B, Swain B, Saha S, Khanra S, et al. Epithelial-to-mesenchymal transition (EMT) and cancer metastasis: the status quo of methods and experimental models 2025. Mol Cancer [Internet]. 2025;24(1). Available from: https://doi.org/10.1186/s12943-025-02338-2
Enderle-Ammour K, Bader M, Ahrens TD, Franke K, Timme S, Csanadi A, et al. Form follows function: Morphological and immunohistological insights into epithelial-mesenchymal transition characteristics of tumor buds. Tumor Biol. 2017;39(5).
Lamouille S, Xu J, Derynck R. Molecular mechanism of epithelial-mesenchymal transiyion. Nat Rev Mol Cell Biol [Internet]. 2014;15(3):178–96. Available from: file:///C:/Users/ASUS/Desktop/Rujukan PhD/EMT/nihms641027.pdf
Ghafoor S, Garcia E, Jay DJ, Persad S. Molecular Mechanisms Regulating Epithelial Mesenchymal Transition (EMT) to Promote Cancer Progression. Int J Mol Sci. 2025;26(9).
Kalluri R, Weinberg RA. The basics of epithelial-mesenchymal transition. J Clin Invest. 2009;119(Jun):1420–8.
Fontana R, Mestre-Farrera A, Yang J. Update on Epithelial-Mesenchymal Plasticity in Cancer Progression. Annu Rev Pathol Mech Dis. 2024;19(1):133–56.
Huang Z, Zhang Z, Zhou C, Liu L, Huang C. Epithelial–mesenchymal transition: The history, regulatory mechanism, and cancer therapeutic opportunities. MedComm. 2022;3(2):1–42.
Li X, Gardner EE, Molina-Pinelo S, Wilhelm C, Mu P, Quintanal-Villalonga Á. Lineage plasticity and histological transformation: tumor histology as a spectrum. Cell Res [Internet]. 2025;35(11):803–23. Available from: http://dx.doi.org/10.1038/s41422-025-01180-x
Gupta PB, Pastushenko I, Skibinski A, Blainplain C, Kuperwasser C. Phenotypic plasticity as a driver of cancer formation progression and resistance to therapy. Cell Stem Cell. 2020;03(24):65–78.
Tam WL, Weinberg RA. The epigenetics of epithelial-mesenchymal plasticity in cancer. Nat med. 2013;19(1):1438–49.
Gonzalez DM, Medici D. Signaling mechanisms of the epithelial-mesenchymal transition. Sci Signal. 2014;7(344):re8.
Buyuk B, Jin S, Ye K. Epithelial-to-Mesenchymal Transition Signaling Pathways Responsible for Breast Cancer Metastasis. Cell Mol Bioeng. 2022;15(1):1–13.
Janiszewska M, Primi MC, Izard T. Cell adhesion in cancer: Beyond the migration of single cells. J Biol Chem [Internet]. 2020;295(8):2495–505. Available from: http://dx.doi.org/10.1074/jbc.REV119.007759
Kumar P, Vishwanath M, Selvaraj S. Tumour Budding with Epithelial Mesenchymal Transition Markers – A Novel Prognostic Tool for Breast Carcinoma. J Midlife Health. 2025;16(2):186–91.
Basu A, Paul MK, Alioscha-Perez M, Grosberg A, Sahli H, Dubinett SM, et al. Statistical parametrization of cell cytoskeleton reveals lung cancer cytoskeletal phenotype with partial EMT signature. Commun Biol. 2022;5(1).
Datta A, Deng S, Gopal V, Yap KCH, Halim CE, Lye ML, et al. Cytoskeletal dynamics in epithelial-mesenchymal transition: Insights into therapeutic targets for cancer metastasis. Cancers (Basel). 2021;13(8):1–27.
Sánchez-Ramírez D, Medrano-Guzmán R, Candanedo-González F, De Anda-González J, García-Rios LE, Pérez-Koldenkova V, et al. High expression of both desmoplastic stroma and epithelial to mesenchymal transition markers associate with shorter survival in pancreatic ductal adenocarcinoma. Eur J Histochem. 2022;66(1).
Hu Q, Wang Y, Yao S, Mao Y, Liu L, Li Z, et al. Desmoplastic Reaction Associates with Prognosis and Adjuvant Chemotherapy Response in Colorectal Cancer: A Multicenter Retrospective Study. Cancer Res Commun. 2023;3(6):1057–66.
Brabletz S, Schuhwerk H, Brabletz T, Stemmler MP. Dynamic EMT: a multi‐tool for tumor progression. EMBO J. 2021;40(18):1–22.
Pal A, Barrett TF, Paolini R, Parikh A, Puram S V. Partial EMT in head and neck cancer biology: A spectrum instead of a switch. Oncogene. 2022;40(32):5049–65.
Chen Y, Ding H, Song J, Gao X. Breast neoplasm epithelial-mesenchymal transition and cytokines: a systematic review. Cancer Cell Int . 2025;25(1).
Yuan J, Yang L, Zhang H, Beeraka NM, Zhang D, Wang Q, et al. Decoding tumor microenvironment: EMT modulation in breast cancer metastasis and therapeutic resistance, and implications of novel immune checkpoint blockers. Biomed Pharmacother [Internet]. 2024;181:117714. Available from: https://doi.org/10.1016/j.biopha.2024.117714
Mohd Sobri SN, Abdul Sani SF, Sabtu SN, Looi LM, Chiew SF, Pathmanathan D, et al. Structural Studies of Epithelial Mesenchymal Transition Breast Tissues. Sci Rep. 2020;10(1):1–16.
Lu W, Chia C, Larby J, Haug G, Hardikar A, Singhera GK, et al. Epithelial to mesenchymal transition (EMT) changes in patients with non-small cell lung cancer (NSCLC). Eur Respir J [Internet]. 62(suppl 67):PA2336. Available from: http://erj.ersjournals.com/content/erj/62/suppl_67/PA2336.abstract
Chang YP, Huang GK, Chen YC, Huang KT, Chen YM, Lin CY, et al. E-cadherin expression in the tumor microenvironment of advanced epidermal growth factor receptor-mutant lung adenocarcinoma and the association with prognosis. BMC Cancer. 2023;23(1):1–11.
Ye Y, Yu S, Guo T, Zhang S, Shen X, Han G. Epithelial–Mesenchymal Transition in Non-Small Cell Lung Cancer Management: Opportunities and Challenges. Biomolecules. 2024;14(12).
Perez-Oquendo M, Manshouri R, Tian Y, Fradette JJ, Rodriguez BL, Kundu ST, et al. ZEB1 is regulated by K811 acetylation to promote stability, NuRD complex interactions, EMT, and NSCLC metastasis. Mol Cancer Res. 2023;21(8):779–94.
Dwi Yanti Fioni Putri, Yenita, Aswiyanti Asri, Tofrizal, Rony Rustam, Husna Yetti. Loss of E-cadherin Expression Stratifies Aggressive versus Non-Aggressive Papillary Thyroid Carcinoma. Biosci Med J Biomed Transl Res. 2025;10(2):361–74.
Xiang J, Lv N, Yin S, Zhao T, Liu F, Cheng L, et al. TGF-β induces EMT in thyroid cancer cells by regulating transcription factors. Thyroid Res. 2025;18(1).
Calangiu CM, Simionescu CE, Stepan AE, Cernea D, Zăvoi RE, Mărgăritescu C. The expression of ck19, Vimentin and e-cadherin in differentiated thyroid carcinomas. Rom J Morphol Embryol. 2014;55(3):919–25.
Montemayor-Garcia C, Hardin H, Guo Z, Larrain C, Buehler D, Asioli S, et al. The role of epithelial mesenchymal transition markers in thyroid carcinoma progression. Endocr Pathol. 2013;24(4):206–12.
Lu J, Kornmann M, Traub B. Role of Epithelial to Mesenchymal Transition in Colorectal Cancer. Int J Mol Sci. 2023;24(19).
Emile MH, Emile SH, El-Karef AA, Ebrahim MA, Mohammed IE, Ibrahim DA. Association between the expression of epithelial–mesenchymal transition (EMT)-related markers and oncologic outcomes of colorectal cancer. Updates Surg [Internet]. 2024;76(6):2181–91. Available from: https://doi.org/10.1007/s13304-024-01865-9
Nie F, Sun X, Sun J, Zhang J, Wang Y. Epithelial-mesenchymal transition in colorectal cancer metastasis and progression: molecular mechanisms and therapeutic strategies. Cell Death Discov. 2025;11(1):1–13.
González-González R, Ortiz-Sarabia G, Molina-Frechero N, Salas-Pacheco JM, Salas-Pacheco SM, Lavalle-Carrasco J, et al. Epithelial–mesenchymal transition associated with head and neck squamous cell carcinomas: A review. Cancers (Basel). 2021;13(12):1–20.
Kadeh H, Saravani S, Moghaddam EM. Immunohistochemical expression of epithelial mesenchymal transition proteins in squamous cell carcinoma of the oral cavity. Iran J Pathol. 2021;16(4):354–61.
Dong D, Zhou Z, Zhu M, Hou Z, Chen M, Gong J, et al. STN1 facilitates metastasis by promoting transcription of EMT-activator ZEB1 in pancreatic cancer. Nat Commun . 2025;16(1).
Sommariva M, Gagliano N. E-Cadherin in Pancreatic Ductal Adenocarcinoma: A Multifaceted Actor during EMT. Cells. 2020;9(4).
Yang J, Liu Y, Liu S. The role of epithelial-mesenchymal transition and autophagy in pancreatic ductal adenocarcinoma invasion. Cell Death Dis. 2023;14(8).
Marrelli D, Marano L, Ambrosio MR, Carbone L, Spagnoli L, Petrioli R, et al. Immunohistochemical Markers of the Epithelial-to-Mesenchymal Transition (EMT) Are Related to Extensive Lymph Nodal Spread, Peritoneal Dissemination, and Poor Prognosis in the Microsatellite-Stable Diffuse Histotype of Gastric Cancer. Cancers (Basel). 2022;14(24):1–15.
Poryazova E, Serteva D, Markov D, Chonov V, Markov G. Expression of Snail and Twist Compared with Clinical and Pathological Parameters in Patients with Gastric Cancer. Folia Med (Plovdiv). 2023;65(3):393–8.
Soukupova J, Malfettone A, Bertran E, Hernández-Alvarez MI, Peñuelas-Haro I, Dituri F, et al. Epithelial–mesenchymal transition (Emt) induced by tgf-β in hepatocellular carcinoma cells reprograms lipid metabolism. Int J Mol Sci. 2021;22(11):1–15.
Wang S, He Y, Wang J, Luo E. Re-exploration of immunotherapy targeting EMT of hepatocellular carcinoma: Starting from the NF-κB pathway. Biomed Pharmacother [Internet]. 2024;174(January):116566. Available from: https://doi.org/10.1016/j.biopha.2024.116566
Pawlicka M, Gumbarewicz E, Błaszczak E, Stepulak A. Transcription Factors and Markers Related to Epithelial–Mesenchymal Transition and Their Role in Resistance to Therapies in Head and Neck Cancers. Cancers (Basel). 2024;16(7).
Cleere EF, Prunty S, O’Neill JP. Anaplastic thyroid cancer:Improved understanding of what remains a deadly disease. Surg [Internet]. 2024;22(1):e48–53. Available from: https://www.sciencedirect.com/science/article/pii/S1479666X23001142
Zhang YC, Zhang YT, Wang Y, Zhao Y, He LJ. What role does PDL1 play in EMT changes in tumors and fibrosis? Front Immunol. 2023;14(August):1–13.